By Joe Gorelick, MSN, FNP-C
With 10 JAK inhibitors now approved by the FDA for use in the US across a range of indications, the significance of the boxed warning for JAK inhibitors to include the risk of major adverse events remains unclear. The lack of clarity with respect to safety is a point of confusion for prescribers and leads to safety concerns in the AD patient population. Another point of confusion within dermatology is that these boxed warnings are based on study data from Non-Atopic Dermatitis patient populations.
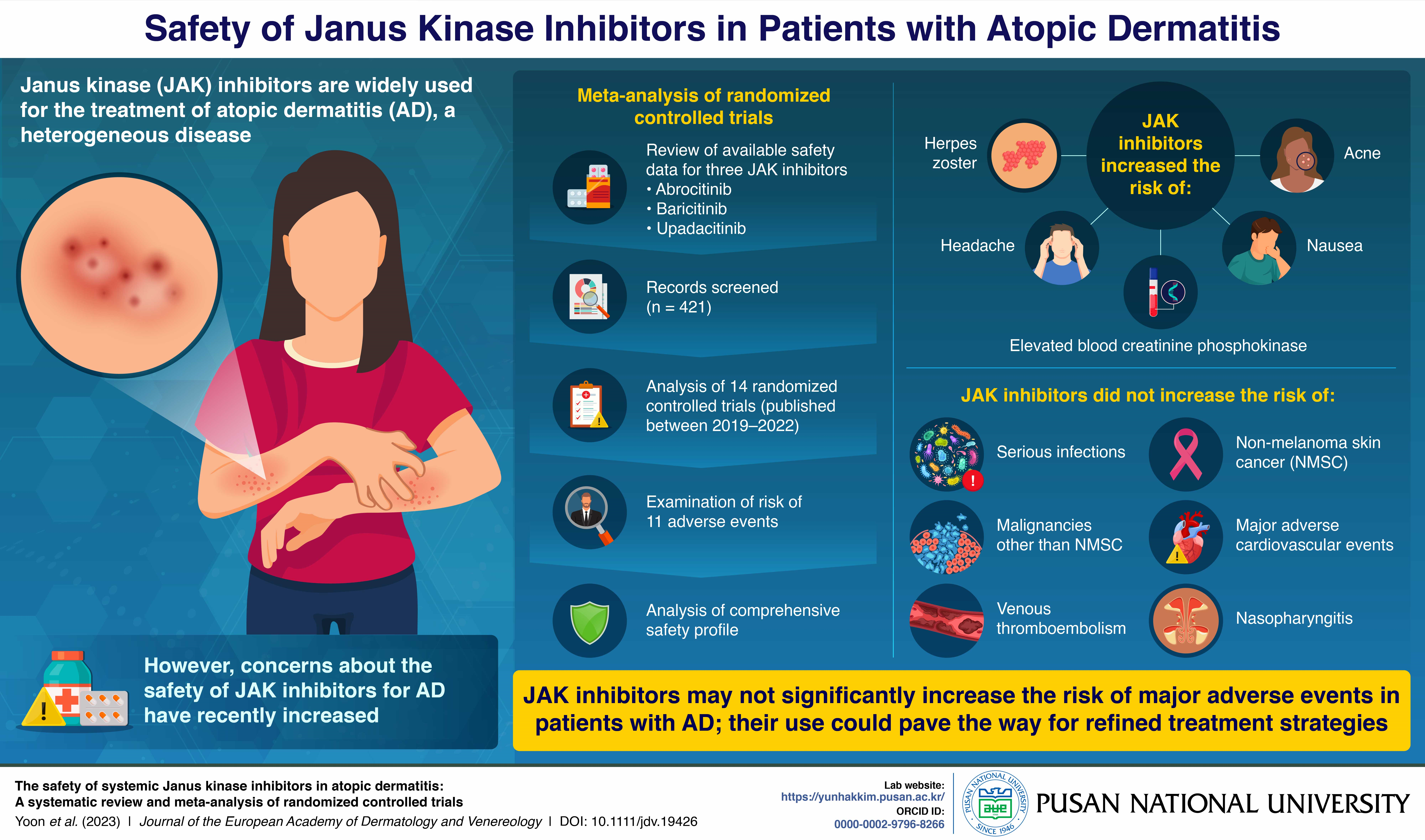
A new meta-analysis from researchers at Pusan National University in South Korea indicates that the incidence of serious conditions did not increase significantly in patients with AD who were exposed to JAK inhibitors.
Researchers conducted an extensive systematic review and meta-analysis of randomized controlled trials (RCTs) involving JAK inhibitors in patients with AD. They identified studies reporting the adverse events associated with JAK inhibitor use from Medline, Embase, Clinicaltrials.gov, among others. They reviewed the available safety data and analyzed the risk of 11 adverse events from 14 RCTs published between 2019 and 2022.
According to the analysis, the relative risk of developing herpes zoster, headache, acne, elevated blood creatinine phosphokinase, and nausea increased among those patients with AD who received JAK inhibitors. However, there was no observed increase in risk for malignancy, venous thromboembolism, and major adverse cardiac events.
The team also assessed risk based on JAK inhibitor type or dose. Among abrocitinib, upadacitinib, and baricitinib (not FDA approved for AD, but approved for AD in Europe, Japan, and Korea), both upadacitinib and abrocitinib were associated with a dose-dependent increase in risk for acne. Higher doses of JAK inhibitors were correlated with an increased risk of acne and elevated blood pressure.
Findings appear in the Journal of the European Academy of Dermatology and Venereology.
Image credit: Yun Hak Kim from Pusan National University, Korea